I mentioned in a previous post that, in my unvarnished opinion, the ACE curriculum was “bad pedagogy and bad science”. I have my reasons for saying this – not the least of which is the fact that I went, for several of my childhood years, to an ACE school.
Let me briefly describe life in the ACE school. The school I attended was a small school, and what passed for learning in that school was to sit in a desk facing a white wall. On the sides of the desk were red and blue dividers to prevent you from looking to the sides. The day consisted mainly of sitting in that little isolated desk and working through workbooks, called PACEs.
If you had questions or needed to take the test at the end of each workbook, you were to raise a flag (either an American or a Christian flag – depending on what you needed), and one of the “supervisors” would come by and attempt to help you. Help was often rather limited, as the supervisors weren’t necessarily experts in any particular area of the curriculum. The supervisors meant well, I suppose, but they were far more concerned with keeping an appearance of order than they were about scholarship.
If sitting at a desk most of the day working through bland workbooks and staring at a blue, a white, and a red wall sounds to you like a lot like an inquisitive child’s vision of hell …. that’s exactly how it felt to me. I would not wish this type of education on my worst enemy or his children. Thankfully, I was eventually sent to a more sane school after four years of this – but I’ve always felt that my four years at an ACE school stunted my intellectual growth. It takes a long time to deprogram yourself of all that nonsense …
At the risk of bringing on some nasty flashbacks to my ACE days, I’ve dug up some samples of the ACE curriculum – so you can judge for yourselves how awful this stuff is.
The lessons start off looking mostly harmless.
Here’s an early sample of Math, for first graders. Counting money is, of course, something you’d want kids to pick up. But the curriculum rapidly goes downhill from there.
Since I’m a teacher of science, I’m going to focus on the science part of the curriculum for now.
Here’s a page from first grade science that describes the taste buds. You’ll notice that the page is as much about thanking God for taste buds as it is about the taste buds themselves. Also notice that the kids are asked to fill in the blanks, with answers that are trivially easy to find in the preceding few paragraphs. While this might not be much of an issue in first grade, the entire curriculum is based on “read and regurgitate” – little if any critical thought is involved.
Here’s another page from first grade science. This page highlights one of the severe failings of the ACE curriculum – it’s more about making kids into fundamentalists than it is about educating kids. Can anyone tell me what this has to do with science, and why it is in the science workbook?
God made all things.
So, all things belong to Him.
All things tell us that God is good.
All things tell us that He is wise and kind.
All things we see tell us that God loves us.
He helps us all day and all night.
He will help us all the time.
God is wise, good, and kind.
The Bible tells us so.
This might be a fine Bible lesson (provided you don’t let the kids hear about Katrina or that tsunami in Asia that killed 200000+ just after Christmas), but it’s in the wrong place.
Let’s move on to third grade science. Here’s what passes for the history of the Earth in the ACE curriculum. The most obvious criticism of this material is that it isn’t science at all – it’s simply part of Genesis in simpler words.
Another criticism of this material is that, again, no thought is involved. For instance, the text says that
There is a band of air which God placed around the earth on the second day.
It then asks the kids to select the best completion to this sentence.
There is a (creation, sand, band) of air around the earth.
Whether you know the real answer or not, only one answer can fit! Lots of ACE questions are this way – even on their end-of-workbook tests. It’s like this at the higher levels, too.
If you have the stomach for it, continue reading the sample third grade science book: here, here, here, here, and here. You’ll find no science, of course. You’ll find only fundamentalism – in big print.
Moving on into the fourth grade, you’ll find that the science ACE is peddling doesn’t get any better.
We use measurement to compare one object with another.
If we want to check or measure our own lives, we compare ourselves only to God. We do not measure up to God because we are sinners.
The curriculum is short on science, and long on fundamentalism. And, like the other pages we’ve looked at, the ACE curriculum relies almost entirely on rote memorization. Science is not viewed as a process of discovery – it’s viewed as a laundry list of facts to memorize. Facts are important, but they’re only part of science.
One thing that I noticed while I was in the ACE school was that the later science PACEs seemed afraid of presenting science. The curriculum was careful to dismiss well-established scientific ideas as “what scientists believe” and cast doubt on established science that might not agree with the ACE authors’ take on the Bible. Take a look at this sample.
Have you ever wondered how many kinds of plants there are? Even scientists do not know for sure. They think there are about 350,000 varieties; however, no one but God knows exactly how many kinds of plants exist in the world.
Sounds innocent (for a religious school) so far, right? Read on, in the ninth grade ACE materials.
Most scientists classify man as a mammal in the phylum Chordata since he has characteristics similar to those of mammals. Man, however, is a unique being with characteristics that he alone possesses. For this reason, we will not classify man as a mammal. Man is not an animal – he is a unique being who was created in God’s image.
Obviously, the ACE curriculum doesn’t teach evolution – the theory that binds biology together. So biology is simply presented as a big dump of largely unrelated information. Much of ACE biology revolves around the classification of organisms. But ACE can’t even give the kids that without screwing it up with fundamentalism!
Finally, we come to tenth grade science. The site I’ve been pulling this material from doesn’t have much in the way of actual content from the science part of the curriculum at this grade level, but what they do provide supports the points I’ve made above. Just take a look at this tenth grade science quiz. For reference, in the tenth grade at the normal high school I went to after escaping from the ACE school, I was taking laboratory-based chemistry and biology courses. The poor ACE kids at that level sit in their cubicles and regurgitate stuff like this:
Special revelation ______________________.
A) reveals God in natural laws
B) is the Word of God
C) teaches man how to know God
D) reveals Who God is
E) B, C, and D
F) A, B, and C
G) all the above
It goes without saying that special revelation is not a scientific concept at all, and has no place in a decent science curriculum. If you click the link above, you can see that the other questions on the page are worded in such a way as to overstate the uncertainties in science. While it’s true that in science all knowledge is provisional, this point isn’t what the ACE curriculum tries to drive home. The ACE point is, plainly put, don’t trust science.
In summary, here’s why i think the ACE curriculum should be put “in the hole”.
- It relies on rote memorization – and only rote memorization – in most areas except some parts of math.
- The assessments are simplistic and don’t involve any sort of critical thought.
- The content is so steeped in fundamentalism that important topics are either left out or distorted. This is especially apparent in science, where the curriculum spends much of its time on theology instead of science.

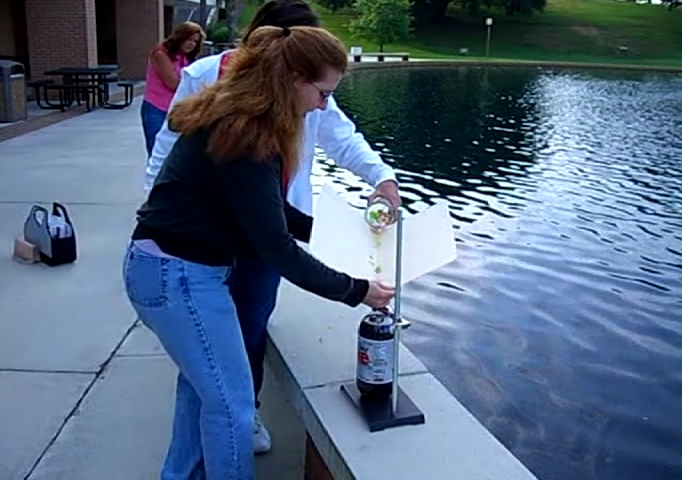
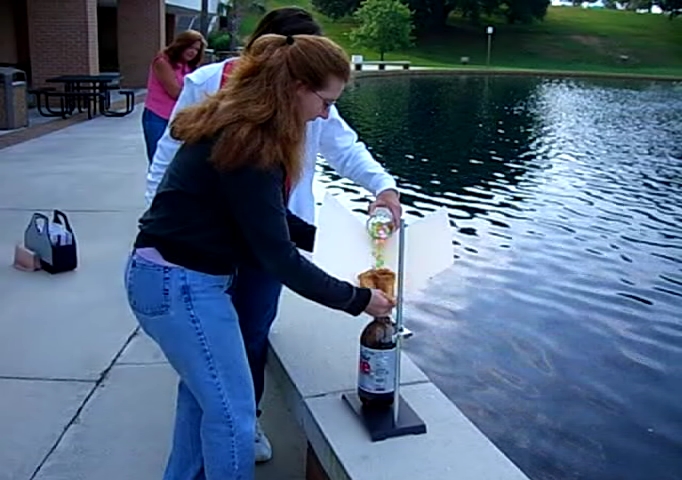
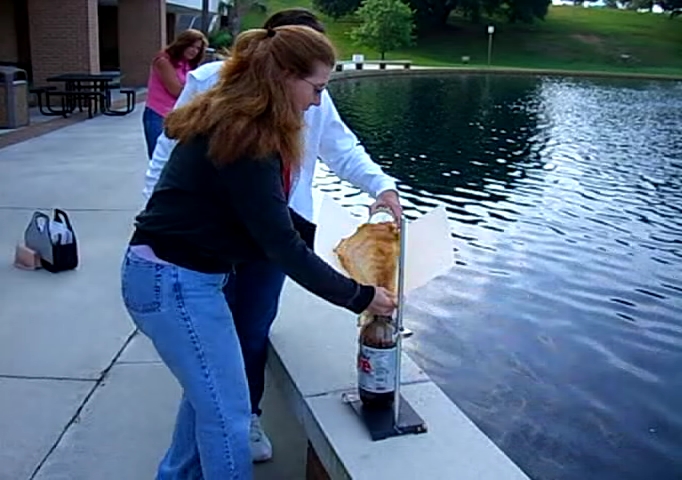
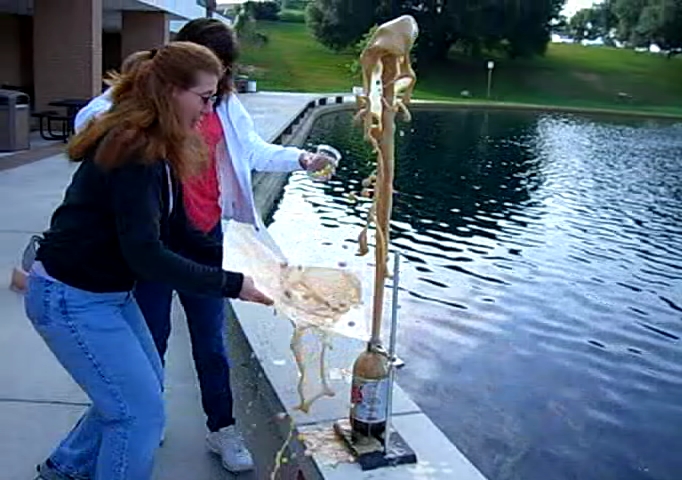

![[Liquid bromine in a beaker]](http://whenchemistsattack.com/wp-content/uploads/2008/01/albr_before.jpg)
![[Aluminum bromide reaction]](http://whenchemistsattack.com/wp-content/uploads/2008/01/albr_still.jpg)
![[More reaction]](http://whenchemistsattack.com/blogfiles/albr_still_2.jpg)
![[FIRE!]](http://whenchemistsattack.com/blogfiles/albr_still_3.jpg)
![[Aluminum burned to a beaker]](http://whenchemistsattack.com/wp-content/uploads/2008/01/albr_aluminum.jpg)
![[Aluminum bromide]](http://whenchemistsattack.com/wp-content/uploads/2008/01/albr_product.jpg)
![[cis-dichloroethene]](http://whenchemistsattack.com/blogfiles/cisdce.png)
![[trans-dichloroethene]](http://whenchemistsattack.com/blogfiles/transdce.png)
![[PIE Logo]](http://whenchemistsattack.com/blogfiles/mind_seige_1.png)
![[Mind seige]](http://whenchemistsattack.com/blogfiles/mind_seige_2.png)